 Evidence uncovered by EnviroReporter.com strongly suggests that these aerosol trails – chemtrails – are “scavenging” deadly Fukushima radionuclides as they settle to earth.
Evidence uncovered by EnviroReporter.com strongly suggests that these aerosol trails – chemtrails – are “scavenging” deadly Fukushima radionuclides as they settle to earth.
Radioactive contamination steaming out the destroyed reactors up into the air, that doesn’t get rained out in storms across the Pacific and North America, stays suspended for a time in the troposphere, the lowest part of the atmosphere. This floating radiation is ripe for chemtrail molecules’ “accumulation mode” in which numerous smaller particles stick to a larger particle until there are so many Fukushima radionuclides stuck to the conglomerated particle that it floats down from the sky.
Whatever the reason for the massive spraying of aerosols in the atmosphere, be they made of aluminum, barium or some other exotic blend of heavy metals or chemicals, these plainly visible gas trails and their resultant cloud cover may be inadvertently nuking us. Fukushima radiation coming down in chemtrail fallout is a situation that even the faceless people behind the spraying in the first place may not be aware of.
According to a 2007 University of Tennessee Department of Nuclear Engineering health physics report, “radioactive particulate matter is carried in air within dust particles.” Once a particle or aerosol is suspended in the air at any elevation it can be removed, altered or destroyed but can’t remain in the atmosphere indefinitely, according to Dr. James Dorsey of the University of Manchester where Dorsey earned a PhD in Atmospheric Physics in 2002. “Clearly the lifetime of any particular particle depends on its size and location,” Dorsey wrote. “Larger aerosol settle out of the atmosphere very quickly under gravity, and some surfaces are more efficient at capturing aerosol than others.”
Aerosols come from being ejected into the atmosphere, like Fukushima Dai-ichi’s continued gas escape of radioactive gas from the destroyed reactors, which joins other “primary aerosols” like volcanic emissions, sea spray, and wind-blown dust. “Secondary aerosols” form as atmospheric gases react and condense and by gas-to-particle conversion as vapors cool. Rainout occurs when an aerosol acts like nuclei for cloud droplets to form around and fall from the sky. Washout indicates an aerosol being knocked out of the sky by a preformed water droplet.
Both rainouts and washouts cleanse the troposphere of significant amounts of Fukushima radiation. The radiation doesn’t disappear, however, and exists as long as its half-life dictates, a half-life being the amount of time it takes for the radionuclide’s mass (weight) to decrease by half. After ten half-lives, a radionuclide’s mass has essentially been reduced to nothing from ionization.
In dry environments, where very small radioactive particles have no effective path to deposition over land or water bodies, nanoparticles join together in a process known as coagulation. These tiny particles, smaller than 100 nanometers, collide into each other due to forces that include electrostatic attraction, phoretic effects like that of heavier particles on lighter ones and transport of a particle due to temperature gradients where the particle moves from a high temperature to a low one. There is also Brownian diffusion which is the effect of thermal random motion on these particles.
Though complicated, these methods are how particles combine and fall to earth. Indeed, the drier the environment, the easier it is for particles to stick together because they don’t have to ‘squeeze’ the water molecules out first to join together. “In the kinetic regime, coagulation is always enhanced due to the absence of viscous forces,” according to Dorsey. That means Fukushima fallout would better bind with spewing aerosol trails better in drier climes like the desert than wet ones.

The actual method of dispersing the aerosols into the atmosphere by jet nozzles also helps the process – the gas literally smacks into the Fukushima fallout and binds to it. “Coagulation is also enhanced in shearing or turbulent flows, as these induce fast relative particle motion,” wrote Dorsey.
Radioactive particulate matter that exploded out of Fukushima the day of the meltdowns and still escaping quickly dispersed over the entire Northern Hemisphere according to an October 20, 2011 study led by the Norwegian Institute for Air Research (NILU) in Kjeller, Norway. The report, Xenon-133 and caesium-137 releases into the atmosphere from the Fukushima Dai-ichi nuclear power plant: determination of the source term, atmospheric dispersion, and deposition, relied on confidential Japanese measurements of Cesium-137 deposition in Japan in 46 of Japan’s 47 prefectures.
 “The plume was also dispersed quickly over the entire Northern Hemisphere, first reaching North America on 15 March,” the report said. (EnviroReporter.com began its 24/7 online Radiation Station Santa Monica the same day.) The study estimated that 19 percent of the total fallout from the disaster’s first five weeks until April 20, 2011 landed on Japan, 79 percent on the Pacific Ocean and “only 2 percent of the total fallout were deposited on land areas other than Japan.”
“The plume was also dispersed quickly over the entire Northern Hemisphere, first reaching North America on 15 March,” the report said. (EnviroReporter.com began its 24/7 online Radiation Station Santa Monica the same day.) The study estimated that 19 percent of the total fallout from the disaster’s first five weeks until April 20, 2011 landed on Japan, 79 percent on the Pacific Ocean and “only 2 percent of the total fallout were deposited on land areas other than Japan.”
Unsound science undoes much of the good work of comprehensive studies with a bias and this report is no exception. Conceptually, it isn’t too hard to figure out that is impossible to estimate these amounts when the meltdowns are ongoing, the melted cores call “corium” unaccounted for and rarely discussed by the reactor owners and Japanese government, and hundreds of thousands of tons of radioactive rubble are being incinerated all over Japan to get rid of the stuff causing a second huge source of emissions. There is no way to quantify that other than to say it is an astronomical amount of goo.
But even mistakes don’t render this report entirely unusable. It reveals that the entire Xenon-133 inventory of radiation was released during the meltdowns, an amount 2.5 times the Xe-133 spewed into the Ukrainian countryside by the Chernobyl meltdown of 1986. Even though the study relied on the June 2011 Japanese government report that first admitted there were three total meltdowns, a report that subsequently was found to have grossly underestimated radiation escaping Fukushima, the NILU study found that at least 42 percent of the amount of Cesium-137 that blew out of Chernobyl had already escaped the Japanese reactor complex hard on the Pacific Ocean. Clearly by now, much more Cs-137 has come out of the battered Fukushima Dai-ichi complex.
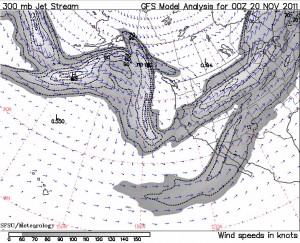
The report said that even with the heavy “scavenging” of Cs-137 by precipitation, the airborne goo had made its way to the Southern Hemisphere. The main brunt was and continues to be north of the Equator. “A first radionuclide cloud ahead of the main plume was transported quickly across the North Pacific at low altitudes in a steady westerly flow and arrived in western North America on 15 March,” the study said. “The main plume was at that time still far from the coast… south of 40 [degrees north], the plume was transported near the surface, while further north it had been lifted to the middle and upper troposphere.”
The troposphere is the Earth’s lowest part of the atmosphere and contains 80 percent of the entire atmosphere’s weight, or mass, and 99 percent of its aerosols and water vapor. What goes up in the troposphere must come down, and fairly quickly, which seems to contradict continued small amounts of radioactive fallout released from the 521 above ground nuclear bomb detonations that took place from 1945 to 1980. Much of that radiation, however, was literally blown into the stratosphere where it still comes down from today. The Fukushima radiation plume, even on the first day of the meltdowns and the explosion of several reactors, did not get propelled into the stratosphere which did, however, limit its vertical height.
 A 1989 EPA radiation risk assessment report showed that “[R]adionuclides, released in the form of particulates or gases, form a plume that disperses down wind. These radionuclides in the air can directly affect people in two ways: through external dose caused by photon exposure from the plume, or through internal dose resulting from radionuclide inhalation. As the airborne radionuclides move from the point of release, they (especially those in particulate form) deposit on ground surfaces and vegetation as a result of dry deposition and precipitation scavenging. Photon radiation from the radionuclides deposited on the ground contributes to the external doses. Finally, small fractions of the radionuclides deposited on plant surfaces and agricultural land enter the food chains, concentrating in produce and in animal products such as milk and meat. Consumption of contaminated foodstuff then contributes to the internal doses of radiation to individuals.”
A 1989 EPA radiation risk assessment report showed that “[R]adionuclides, released in the form of particulates or gases, form a plume that disperses down wind. These radionuclides in the air can directly affect people in two ways: through external dose caused by photon exposure from the plume, or through internal dose resulting from radionuclide inhalation. As the airborne radionuclides move from the point of release, they (especially those in particulate form) deposit on ground surfaces and vegetation as a result of dry deposition and precipitation scavenging. Photon radiation from the radionuclides deposited on the ground contributes to the external doses. Finally, small fractions of the radionuclides deposited on plant surfaces and agricultural land enter the food chains, concentrating in produce and in animal products such as milk and meat. Consumption of contaminated foodstuff then contributes to the internal doses of radiation to individuals.”
While the EPA summation is accurate and its information may seem obvious to experienced fallout experts, aerosol trails streaked across the skies certainly weren’t taken into consideration because they didn’t exist in the multitudes that they have from at least the mid-1990s. The term “chemtrail” was first coined by journalist William Thomas in 1997. Nevertheless, what the extensive EPA analysis confirmed was that radionuclides like Cesium-137 released like those that have been from Fukushima Dai-ichi behave in identifiable patterns.
“As radionuclides in the plume are dispersed, their activity is depleted by dry deposition and precipitation scavenging,” the study noted. “Depletion due to deposition generally does not cause more than half of the released activity to be removed at a distance of 80 [kilometers]. Depletion by precipitation scavenging occurs only during periods of precipitation.”
Nevertheless, there are other ways to get the Fuku-goo cloud to fallout. A new NILU study adds cloud scavenging and dry deposition to the mechanics of getting goo to ground. The Norwegian team worked with the Central Institute for Meteorology and Geodynamics in Vienna, Austria to produce a May 14 report for the European Geosciences Union called Atmospheric removal times of the aerosol-bound radionuclides 137Cs and 131I during the months after the Fukushima Dai-ichi nuclear power plant accident – a constraint for air quality and climate models.
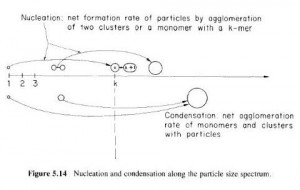
Though once again hampered by mistakes and misassumptions, the report is certainly useful. It shows how Fukushima Cesium-137 in the troposphere is captured and coagulated in “accumulation-mode” before falling out. “Caesium-137 (137Cs) and iodine-131 (131I) are radionuclides of particular concern during nuclear accidents, because they are emitted in large amounts and are of significant health impact,” the study reported. “137Cs and 131I attach to the ambient accumulation-mode (AM) aerosols and share their fate as the aerosols are removed from the atmosphere by scavenging within clouds, precipitation and dry deposition.”
The May 14 NILU study found “For the emissions from the Fukushima Dai-ichi nuclear power plant accident in March 2011, there is direct evidence that the 137Cs was attached to aerosols in the size range 0.1–2 μm [micrometer – 1 millionth of a meter] diameter,” and that “Once attached, 137Cs shares the fate of these aerosols, which grow by coagulation with other particles during transport … and are removed by wet and dry deposition. Thus, the removal rates of 137Cs should be representative for the AM aerosols in general.”
The study’s modeling, while wanting, relied on an impressive set of data from 60 particulate monitoring stations worldwide that delivered data to the International Data Centre of the Preparatory Commission for the Comprehensive Nuclear-Test-Ban Treaty Organization in Vienna, Austria. The radiations stations have high-volume aerosol samplers that test 20,000 cubic meters of air over a 24 hour period collecting particulate matter. High-resolution germanium detectors (of the sort that EnviroReporter.com is seeking to obtain for Radiation Station Santa Monica) then identify the radionuclides present at even low concentrations.
While NILU examined both Iodine-131 and Cesium-137, the former has a half-life of 8.02 days and, absent continuing nuclear fission at the meltdowns site which is still a constant threat, has effectively ionized itself out of existence. The Cs-137 is an entirely different matter since it stays in the environment for centuries and has entered the food chain as previously reported in this article. “[Cesium-137] is released both as gas and in particulate form,” the study said. “The gaseous release fraction is typically as high as the particulate fraction. During transport, there is an exchange between the gas and particle phases.”
Using co-located measurements for radionuclides, NILU determined that the removal times for Cs-137 from the atmosphere range from 8.8 to 18.1 days, more than enough time for it to travel to North America and beyond.
 “The aerosol lifetimes vary regionally and are generally longest in dry or cold regions” the report stated which is bad news in the usually dry areas of the West and Southwest United States like, for example, Death Valley. With the advent of aerosol spraying in the national park as witnessed by this reporter and his editor (and wife), barriers to the Fukushima goo-laden jet stream moisture needed previously to create fallout through wet deposition have been inadvertently overcome as chemtrails scavenge the Cs-137 creating fallout in these arid regions. EnviroReporter.com has seen these aerosol trails spread out in Death Valley and create the sort of bluish haze one associates with the polluted climes of Southern California.
“The aerosol lifetimes vary regionally and are generally longest in dry or cold regions” the report stated which is bad news in the usually dry areas of the West and Southwest United States like, for example, Death Valley. With the advent of aerosol spraying in the national park as witnessed by this reporter and his editor (and wife), barriers to the Fukushima goo-laden jet stream moisture needed previously to create fallout through wet deposition have been inadvertently overcome as chemtrails scavenge the Cs-137 creating fallout in these arid regions. EnviroReporter.com has seen these aerosol trails spread out in Death Valley and create the sort of bluish haze one associates with the polluted climes of Southern California.
Plus, the longer the Cs-137 can literally hang around the worse it is because it can concentrate further in a stationary high-pressure zone upping the possibility of being scavenged by an aerosol trail and any resultant man-made cloud particulates. NILU does admit Fukushima Cesium-137 hang time could be longer. “[T]he comparison between our results and modeled aerosol lifetimes may still indicate that the lifetimes in the aerosol models are too short,” the report concluded. “Our results are highly sensitive to possible late emissions of radionuclides. However, there is no evidence for such late emissions, neither in our data nor in the existing literature on the FD-NPP accident.”












FABULOUS & METICULOUS REPORTING — FINALLY!!! WE APPLAUD YOU!!! YOU ARE BRAVE + VERY WELL-INFORMED! AND ABOVE ALL, YOU ARE A VERITABLE SCIENTIST & INVESTIGATIVE REPORTER!!! THANK YOU, SPECIALLY FOR CONFIRMING MY SIGNIFICANT FEARS — IN AN EFFORT TO PROTECT MYSELF AND HUMANITY FROM THIS ONGOING MASS-EXTINCTION EVENT! …THE ONLY REALITY YOU HAVEN’T YET RELATED, IS THE STARVATION AND MASS DIE-OFF OF SEVERAL “THOUSAND” MARINE SPECIES DUE TO FUKUSHIMA RADIONUCLIDE + CURRENT UPWELLING OF THE ENTIRE WEST COAST OF U.S., CANADA (ESPECIALLY VANCOUVER), AND ALASKA + A-L-L WEST COAST FOODSTUFFS GROWN, ESPECIALLY ORANGES, MUSHROOMS, LETTUCE, SPINACH, RADISHES, CUCUMBERS, PISTACHIOS, ALMONDS, STRAWBERRIES, AND SUMMER + WINTER SQUASH VARIETIES!!! …WHAT IS LEFT TO EAT?? (FISH, MEAT, MILK!, + WATERSHEDS ARE ALSO UNSAFE FOR HUMAN + ANIMAL CONSUMPTION!). THE “DENIAL FACTOR” ESPOUSED BY THE NUCLEAR INDUSTRY, AND “SABOTAGE” OF ALL RELIABLE INFO. IS TRULY STARTLING, AND FURTHER ADDS TO THE CHAOS THAT WILL ERUPT WHEN ALL LIFE WILL STARVE TO DEATH, AND WORLD AGRICULTURE FAILS… MOST NOTABLY IN THE NEXT (2 -3) YEARS… WHAT SUBTERFUGE WILL THE “EXPERT LIARS” FEED US THEN?!?! MY (25) YEAR STOCK INVESTMENTS MIGHT AS WELL BE USED UP WITHIN (3) YEARS… THE CASH AND CURRENCIES WON’T HAVE ANY FOOD TO BUY … THE AVARICE OF A FEW THOUSAND MEN OVER (50+) YEARS WILL TOTALLY DECIMATE THE ENTIRE SPECIES OF LIFE… MARS, HERE WE FOLLOW TO LOSE OUR PLANET AS WELL… I AM TRULY OUTRAGED AT THEIR DENIAL, LIES, ARROGANCE, AND PLUNDERING OF THIS ONCE-BOUNTIFUL PLANET… LET’S ROUND THEM UP AND TOSS THEM IN THE SPENT FUEL POOLS THEY DENY ARE COLLAPSING — CREATING A CRITICALITY EVENT THAT WILL TORCH THE ATMOSPHERE AND MANKIND IN A MATTER OF MONTHS. …APOLOGIES TO THE CREATOR, IF YOU CAN STILL BELIEVE…
What’s the latest update on radiation levels and what can we do about it? There’s a lot of talk about the issues, but not enough about the solutions!
@Carol: You’d be doing yourself a big favor if you looked at our site’s coverage before becoming frustrated. It’s up to you how much you know about the ongoing triple meltdowns spewing millions of tons of extremely radioactive water into the Pacific Ocean and invading the marine flora and fauna. But here’s a good place to start: Fukushima – The Perfect Crime?
OK, so we Know about it now… but are there any Updates? What is Happening NOW??? I’m quite Certain it has NOT been Cleaned UP! Why are we continuing to Allow them to get away with Poisoning our Planet??
Here we are in 2014 and Fukushima radiation has had its way with countries other than the U.S. See:
http://businessinsider.com.au/the-serious-biological-effects-of-fukushima-ratiation-on-plants-insects-and-animals-is-slowly-being-revealed-2014=8
Get out of the Northern Hemisphere ASAP. As an open and modern society Chile is a top contender as it is also one of the most developed countries in South America with a growing expat community. It is surprisingly cheap and easy to move to and if you buy land you are automatically granted permanent residency.
I am DISGUSTED that the Japanese Govt. has not safeguarded (if that is possible) the worlds population by stopping any further meltdown of this plant. Furthermore, dumping this stuff into the ocean is outrageous. I understand it is life threatening to go there but if we can send a man to outer space we can certainly provide clothing and breathing apparatuses to safeguard the workers. There is no reason that this meltdown cannot be contained.
As far as the Chemtrails goes: they have stepped up the spraying in my area. So much so that I rarely go outside anymore. I can taste the stuff and smell the stuff (hot burned dirty metal) in the air. This is all part of Agenda 21. Of this I am convinced. The govt. has underground bunkers built and fully stocked for their survival. Reading the current laws on the books I discovered that the US Govt. can legally spray anything they want onto the American populace (including biological weapons) through a little thing called implied consent. If no dissent is legally voiced consent is implied and given. In order for it to be successfully challenged it must be done through the court system. An environmental attorney is needed. If there is one out there willing to take the case and see it through the courts both national and international it could be stopped. Furthermore the Japanese Govt and the owners of the now defunct nuclear power plant need to be sued. Money won’t fix the disaster they are all creating. However if you break them all financially this nightmare could potentially begin to heal the earth and her inhabitants through Cleanup efforts and through research to heal our contaminated environment. If enough people join together worldwide to deal with the few that choose to perpetuate this violence on our worlds population they wouldn’t be able to continue the lie. They would be outnumbered and have to admit their behavior and the populace would demand that it be stopped. Thank you for your article!
Considering the state of the oceans presently… where does one obtain Chlorella from these days? Seems all sources lead back to Fukushima country or thereabouts that are too close for comfort.
Fukushima Radioactive Contamination Symptoms Research
http://frcsr.blogspot.com/2012/09/10-august-2012.html
“Reports of tumors have begun to come in. There has been a rapid increase of endocrine symptoms, which might be a preliminary stage before tumors appear. In addition, there have been multiple reports of pediatric thyroid cancer from the Kanto region and the Metropolitan area, 250 km away from the Fukushima Dai-ichi nuclear power station.”
“The central nervous system effects include such symptoms as lassitude with fatigue, memory decline/loss, forgetfulness, agraphia, loss of mental calculation, motor dysfunction such as falling, but it is difficult to collect data because they may not be aware of changes in their own characteristics other than what they can perceive themselves for self-reporting.”
@ Michael – In this comprehensive article “No Place to Hide”, you astutely suggest that we get our hair tested for radioactivity so that we can know if we have radiological contamination in ourselves.
I have just read today’s posting in Fukushima Diary that hair was tested from a barber shop in Fukushima and was found to be more radioactive than food! They found combined Cesium 134 and Cesium 137 at levels over 100 Bq/kg.
Sampling date : 10/25 ~ 11/9/2012
■ Test 1
Time : 1,800 seconds
Cs-134 : 59.89 Bq/Kg
Cs-137 : 50.91 Bq/Kg
■ Test 2
Time : 12,000 seconds
Cs-134 : 54.88 Bq/Kg
Cs-137 : 60.62 Bq/Kg
http://fukushima-diary.com/2012/11/human-hair-more-contaminated-than-food-over-100-bqkg-from-hair-of-fukushima-citizen/
I’m wondering if anyone has found a lab that will test hair for radioactivity? The labs are closed today but I will make calls tomorrow and get back to you. Your brilliant idea, Michael, needs to be followed up and acted on.
When LA Weekly‘s Adam Gropman, taking time off of riding buses to catch snippets of twisted conversations to mash into mindless pulp for us to read in the paper, bagged on chemtrails with “You see ordinary contrails; they see poisonous ‘chemtrails'” I thought I would choke from the shame of being a contributor to the paper. I mean, of all days – today – when jets are spurting out little chemtrails over the Los Angeles Basin, it is patently clear these aren’t contrails. Of course, this comes from a newspaper where one of the editors said radiation from Fukushima wasn’t more harmful than getting your arm sunburned while driving!
Regardless, there is now a new website called Los Angeles Skywatch where you can see what’s in Los Angeles chemtrails. The results are astounding.
Now, what’s missing from these results? Fukugoo in the form of fallout radionuclides brought to ground and sea by this outrageous spraying that has even ‘alternative media’ stone-stupid.
If it’s not right to be afraid of radiation, well then Steve ought to go to Japan where workers cannot get within 100 feet of the hot reactors and fuel pools. Since he is not scared of it, and all..
@Liz – Butler Henderson
Thanks for this information – very useful.
You can buy zeolite a lot cheaper than the liquid zeolite seen most widely advertised on the net. Liquid zeolite is expensive and I don’t know how effective this would be at taking radiation out of your system. At Chernobyl zeolite was ground and baked into bread and cookies. The zeolite traps the radiation and then you eliminate it out of your body. The zeolite remains radioactive this is why a method for decontaminating soil must be invented. If you just add it to the soil it will capture the radiation but then it remains in the soil. The plants, hopefully, wouldn’t uptake the radiation, however, you still have irradiated soil.
For consumption I would strongly advise you purchase zeolite from the southern hemisphere and Australia has some good stuff. Buy the ground zeolite; put it in capsules and take daily.
@BluesTeaCha: Cloud seeding (aka weather modification) has been ongoing in California since the 1950s. A must read is the document “OPTIMIZING CLOUD SEEDING FOR
WATER AND ENERGY IN CALIFORNIA” published in 2007 by the Gov. Schwarzenegger’s office that details some of the history (See: http://www.energy.ca.gov/2007publications/CEC-500-2007-008/CEC-500-2007-008.PDF).
I also found a current state budget for the cloud-seeding programs that includes Santa Barbara County as part of the effort. I suspect this project has been greatly expanded as a climate-change mitigation effort but either from a national budget or a budget area that lacks the kind of transparency that the California report seemingly provides.
Here’s an excerpt from the introduction of the above-linked document:
Operational weather modification (cloud seeding) has been conducted in California since the early 1950s—one of the longest records of seeding in the world. Cloud seeding has been conducted in many areas, but the most continuous programs have been in winter over the Sierra Nevada. This seeding has been intended to augment snowfall and snowpack. The additional snowpack melts and runs off, providing more water for various uses such as hydroelectric power, agriculture, municipal and industrial needs, recreation, and endangered species habitat. The California Department of Water Resources (DWR) has conservatively estimated a 4 percent annual precipitation increase attributable to the combined state seeding projects.
Interesting writing. Thanks.
If the contrails began in 2010, then they are probably not related to Fukushima. It would be interesting, however, if the military had taken advantage of the first opportunity since Chernobyl 1986 to test methods to precipitate radioactive nuclides out of the atmosphere over deserts rather than over areas of high rainfall (and consequently high food production) where the fallout would naturally precipitate. Weather or climate modification is possible, but it’s more likely to be military jets doing something such as training pilots. The contrails would have unusual characteristics as the (very high) altitude or (high-velocity) propellants are different from those of commercial airliners.
@amravati – You asked if there is anything you can take if you think you have ingested radioactivity. I am presently writing a short manual on how to be prepared for a nuclear disaster. One section is called “Supplements to have on hand to protect yourself and combat radiation” It includes 36 supplements I have learned about. I’m just copying from the draft of the manual for you (with apologies to Michael for taking up so much space)
The top 7 are:
1. Potassium Iodide (Amravati – this will not help if you think you’ve already ingested the radioactive Iodine – it needs to be taken before the plume hits)
Arnold Gunderson, now, 16 months after the event at Fukushima Diiach, has said that citizens on the west coast of North America should have taken Potassium Iodide tablets.
Potassium Iodide will saturate the thyroid glands giving the radioactive Iodine 131 no place to land. So it’s a good idea to have Potassium Iodide Tablets in your anti-radiation arsenal. Dr. Caldicott says if there is a meltdown and you know the plume is coming (and you will if you keep yourself informed), then take one tablet before the cloud reaches you. She says one tablet is enough to super saturate the thyroid gland so you won’t get thyroid cancer.
An important note is that potassium iodide is a solution only for blocking the uptake of radioactive Iodine 131. There are several hundred other isotopes released in a nuclear meltdown.
One card of IOSAT tablets has 14 tablets on it and costs $7.45. I ordered mine online from Amazon.com
2. Zeolite binds radioactive isotopes. I have heard both Arnie Gunderson of Fairewinds and Associates and Levy HaLevy of Nuclear Hotseat recommend Zeolite. If you think your fruits and vegetables have been contaminated with radioactivity, you can add liquid zeolite to your filtered vegetable washing water, wash your food well in this solution, and then rinse with filtered water.
I found Liquid Zeolite from the Liquid Zeolite Company. LiquidZeolite@AOL.com One bottle was $36.00
3. Pectin I have seen several documentaries on Chernobyl and I have read the book “Chernobyl – Consequences of the Catastrophe for People and the Environment.” Published by the National Academy of Sciences. All these resources have mentioned that in the contaminated areas, Apple Pectin is given to people to help decrease the levels of ingested Cesium. Pectin chemically binds cations such as Cesium in the gastrointestinal tract and thereby increases fecal excretion. Researchers have concluded that adding pectin to the food of inhabitants of the Chernobyl-contaminated regions promotes an effective excretion of incorporated radionucllides.
I bought Apple Pectin through Amazon.com from Natural Vitamin Direct – one 90 capsule jar is $13.65.
4. Vitamin C has been shown to be protective of radiation damage. According to Dr. Russell Blaylock, some research has “shown substantial reduction in damage to bone marrow cells and thereby lower one’s risk of developing leukemia or severe bone marrow depress (which can result in fatal infections)”.
Recently Dr. Michael Nobel has recommended Pro Pectin http://www.propectin.com/ but I haven’t bought that yet so I do not know the cost.
5. Calcium supplements are apparently a good idea so that your bones are full of calcium. Radioactive Strontium 90 mimics calcium and goes right to your bones if there is room for it. So if you are deficient in calcium the Strontium 90 will get into your bones and cause bone cancer.
I have bought SISU brand Calcium and Magnesium 2:1 with vitamin D at my local health food store.
6. Vitamin D
vitamin D – D3 (also known as calcitriol) – may offer protection against a variety of radiation-induced damages, including even those caused by background radiation or a low-level nuclear incident.
The protective mechanisms are so strong that researchers suggested vitamin D3 should be considered among the prime (if not the primary) non-pharmacological agents to protect against sub-lethal low radiation damage and, particularly, radiation-induced cancer.
Note: Magnesium allows Calcium to get into cells and thus helps fill the bones with Calcium so that Strontium 90 cannot get in.
I have SISU brand Vitamin D3 1000 mg tablets and I take 2 a day but you need to research and determine how much is right for you.
7. Bentonite Clay
Absorbs radiation. I have found both an edible form ($9.89 for ½ pound from Harmonic Arts) and a type that you pour into a bath to soak in and detoxify yourself from ProMedics Neutraceutical Ltd. Vancouver, B.C.
Those are the top 7 supplements to have in your arsenal. There are many more supplements that I have read about but have not yet bought and added to my nuclear preparations. It all takes time and money to research products and order them. Possibly with more time I will add some of the following 29 supplements to my anti-radiation collection. I recommend that you study this issue, start your collection and make your own decisions.
To read more on the other 29 supplements, for $10 you can download Dr. Russell Blaylock’s Nuclear Sunrise .
http://www.blaylockwellnesscenter.com./Literature_Pg.2.html
a. Ginkgo biloba – radioprotective by free radical scavenging
b. vitamin E – radioprotective by free radical scavenging
c. carotinoids – radioprotective by free radical scavenging
d. echinacea – inhances immunity
e. IP-6 – inhances immunity
f. beta 1,3/1,6 D-glucan – inhances immunity and protects against radiation
g. WR-2721 (chemical radioprotectant) – increases effect of beta 1.3/1,6 D-glucan
h. curcumin (flavinoid) – inhances immunity
i. (flavinoid) – inhances immunity
j. orientin (flavinoid) – protects against radiation damage
k. vicenin (flavinoid) – protects against radiation damage
l. Panax ginseng –(herb) – inhances immunity
m. ProstaglandinInhibitors such as Advil – protects bone marrow from radiation damage
n. Garlic – protects against whole body irradiation by gamma rays
o. Chlorella (algae) – protects from sublethal doses of gamma radiatioin
p. Selenium – optimum dose 200 micrograms per day
q. Melatonin – radioprotectant properties
r. Alpha-lipoic acid – radioportectant
s. curcumin (flavinoid) – radioprotectant fo all tissues, cells and organs
t. ginger – anti-inflammatory and radioprotectant
u. zinc (mineral) – reduceds radiation-induced elevation of lipid peroxidation
v. Folate, methylcobalamine, niacinamide and vitamin B6
– all act in concert to protect and repair DNA. Dr.Blaylock says the does are 800 micrograms for folate, 5000 micrograms for methylcobalamin, 500 mg for niacinamide and 50 mg for vitamin B6 daily. Do not take iron with these as iron increases free radical generation.
w. Rosemary extract – protects against radiation injury
x. Green tea extract – applied to skin and taken orally protects against UVA/UVB radiation to the skin
y. Resveratrol – caner-preventing compound and radioprotectant. Dr. Blaylock recommends 200 mg twice a day between meals.
z. Four other supplements I have read about and you can study (Not discussed by Dr. Blalock)
– Boron
– used to capture radioactivity on soils, gardens, orchards, etc. It also can be safely ingested by humans and animals. Boron will accept radiation and ionize it within our bodies, after which our bodies will safely excrete the boron and radioactivity.
– Spirulina
– a blue-green algae – might be another useful alternative to protect against the effects of radiation. Spirulina was actually used to treat children exposed to chronic low levels of radiation after the Chernobyl nuclear disaster. It enhances the immune system.
– Organic Turmeric
Reduces damage from both external radiation exposure and ingested radionuclides. by reducing oxidative stress to cellular structures, particularly DNA.
(note: it must be certified organic to ensure it has not been gamma irradiated (ironic?) with radioactive cobalt-60 in the USDA-approved process known as “cold pasteurization.” Conventional spices are commonly exposed to as high as 30 Kilograys of gamma radiation, or the equivalent of 990 million chest x-rays worth of radiation. The irradiation of herbs produces formic acid, formaldehyde and unique radiolytic byproducts with carcinogenic properties.
– Whey protein
Protects against absorbing radioactive minerals.
GreenMedInfo.com has a Radiation Disaster Associated Toxicity page which has indexed 146 substances which may have radioprotective properties.
@SBLocal: “Neil” forgot the saying ‘better to be thought a fool than to open one’s mouth and prove it.’ His first proclamation that the Inspector Alert nuclear radiation monitor is the giveaway: of course it can detect alpha and beta radiation. As for the other claptrap “Neil” offers, we included this comment to remind you all that this entrenched ignorance and malevolence is the province of pro-nukers who still can’t see the folly of nuclear power which has so devastated Japan. Remember, however, that these anonymous voices of the glowing status quo are still stronger than yours which demands a redoubling of efforts if we are ever to stuff the nuclear genie back in its bottle.
@Neil – “Stop raising the fear levels of people.” How about “Stop ordering others to conform to your stolid framing of the current situation”? Do you honestly believe that it isn’t obvious you’ve consumed the kool aid and/or are on the kool aid manufacturer’s payroll? Enough said. I won’t let you further waste my time.
I am wondering what people can do if they believe they may have ingested contaminated foods. Are you aware of any ‘cures’ , practices or substances which can help them. Might also be useful to mention symptoms to alert people. I live in the UK and try to eat local produce as much as possible so am not concerned on my own behalf. People should be warned not to go out in rain which is likely to be contaminated in the areas where this is happening as to get it on their skin would be very detrimental. Perhaps the Government should be issuing fact packs to people with preventative and curative measures.
I doubt the radiation meter, can measure both Alpha and Beta radiation. No doubt there is fallout from Japan, but stop raising the fear levels of people. If all the electricity the world has used that has been generated by nuclear power were generated by other methods. The planet would be much worse off for it. Windmills and solar panels have their place, but will not even come close to meeting global demand. They still require back up, which means fossil fuels. China has numerous Nuclear Plants under construction. Perhaps that is something to be concerned about.
@ Micheal, Susan and Severin
Thanks for being so polite and informative to a most likely one time commenter Steve coats.
Steve says, “I got out of the [Nuclear] industry years ago due to the irrational fear people have of radiation.”
Maybe you should rethink that one Steve.
It’s NOT irrational, it’s that we have figured out ‘the lie’ that Nuclear Power is NOT ‘safe’.
(never could be, never can be)
Unfortunately, Fukushima Daiichi’s 3 meltdowns and continuous radioactive contamination of our air, waters and soil (food), prove that point.
Nuclear Power is a travesty against all innocent living things!
Quote: “I have been following this accident and so far there are NO deaths or significant injuries from radiation in this accident.”
Here is proof that this statement is totally incorrect.
I suggest you all use this document as one of your resources to prove to those who say “No one has died from the Fukushima Nuclear disaster”, they are totally wrong.
This occurred in the first few days of the disaster. In this correspondence between the NRC and the Japanese, the Japanese admitted to 5 people having received lethal radiation doses.
http://enformable.com/wp-content/uploads/2012/01/Pascarelli-Robert.png
@Steve coats If you truly have been educated in health physics, you should be ashamed of yourself for being unaware of what other health professionals already know. Manmade radioactive nuclides are deadly to human beings. They damage our DNA and, when ingested or inhaled, the damage just keeps getting worse. There is a cancer epidemic in full swing Steve. To compare the manmade radioactive poisoning of our water, air and food supply to car accidents is an analogy so false as to draw this reader to wonder about your actual level of education.
@Steve coats Michael Collins is absolutely right that there is NO safe level of radiation.
Read “Chernobyl – Consequences of the Catastrophe for People and the Environment” by Yablokov, Nesterenko and Nesterenko and published by the National Academy of Sciences. There are over 30,000 publications in the scientific literature and millions of documents on the consequences of the catastrophe. This book reflects more than 5000 of those publications.
If you have the stomach for it, watch “Chernobyl Heart and the Children of Chernobyl” http://www.youtube.com/watch?v=8ujAG_Ofj4M
And read “Killing our Own–The Disaster of America’s Experience with Atomic Radiation” http://www.ratical.org/radiation/KillingOurOwn/
Please update your knowledge base with these references.
“Deadly and dangerous” are completely appropriate words when it comes to nuclear power.
The Japanese government is covering up the health effects of the ongoing Fukushima accident. Hopefully brave scientists and physicians will research and publish the true data for japan the say they have done for Chernobyl.
The only hope for our planet is for the people of the world to learn the truth about nuclear power and put all their energies into closing down all the reactors.
Thank goodness we have Michael Collins who is tirelessly reporting and letting people know about deadly and dangerous nuclear power.
@Steve coats: Utter tosh. No amount of low-level radiation exposure is safe. In late July 2005, the prestigious National Academy of Sciences came out with a milestone report that confirmed this. “The scientific research base shows that there is no threshold of exposure below which low levels of ionized radiation can be demonstrated to be harmless or beneficial,” said Richard R. Monson, the panel chairman and a professor of epidemiology at Harvard’s School of Public Health.
I am am health physicist and have worked at a nuc plant (in charge of rad protection) . I got out of the industry years ago due to the irrational fear people have of radiation. While this article may show some facts, it is very predjudicial. Words like deadly and dangerous do not belong in it. There is substantial evidence now that confirms that low levels of radiation can be beneficial( much higher than anyone received in japan). I have been following this accident and so far there are NO deaths or significant injuries from radiation in this accident. We are close to a nuclear war in the middle east, largely because the development of nuclear power has been virtually stopped. If you want to panic, look at real risks such as blocking safe clean power sources and the 40,000 or so people that we kill in cars every year.
Thank you Mr. Collins for the excellent presentation of your great research. In my opinion your work seems like it’s the best available to the public. It’s too bad that our criminal bankster government works so hard to keep us ignorant.
Nuclear waste dumped at “SEA”
Ocean disposal of radioactive waste:
Status report: http://is.gd/tiugbX
Note it is also been dumped around California, for all to enjoy!
Maybe that is now also part of our Background readings!
The Trial Of Minoru Tanaka: The high cost of investigative journalism in Japan & “the nuclear mafia http://www.japansubculture.com/?p=5397
Why information about nuclear reactors does not appear in MSM, sounds familiar!
Two true stories from personal experience:
In first or second grade (about ’75)in Santa Barbara I was taken on a field trip to Rincon to the offshore oil facility island. I have no idea what it’s used for now or then, but at that time it had an “educational” kelp holdfast tank. We were told by “educators” there that during the Santa Barbara oil spill of ’69 the mollusks merely closed their shells and the sea birds flew away; as a result no animals were harmed by the oil spill. Fast forward to UCSB summer school class probably ’89 — “Problems With Energy” taught by Dr. Hal Lewis; the guy who studied under Robert Oppenheimer and who recently resigned from the American Physical Society in opposition to the “global warming scam”. He taught us in that class that nuclear power was absolutely safe beyond doubt. We took a field trip to Diablo Canyon. The class was a platform to extol nuclear power. There was no critical or opposing view offered in the class and it was weird beyond words. Thank you, “Alli”, for the opportunity to tell these stories.
But that was Plutonium in that reactor. Only ONE use for it. Who were they going to sell it to?
http://www.fas.org/sgp/othergov/doe/lanl/pubs/00818005.pdf
Lisa in Wisconsin,
Here are two free guides that can help you.
Geiger counter use guide
http://technologypals.com.au/wp-content/uploads/2012/03/Using-a-Geiger-Counter-to-test-food-for-Radioactive-Contamination.pdf
DIY Food and environment testing lab set up free guide
http://sccc.org.au/wp-content/uploads/2012/06/How-to-set-up-a-home-or-community-food-testing-lab-for-radioactive-contamination.pdf
Do more research, these are just basic guides to get you started. Some radioactive contamination can not be detected without some very sophisticated equipment, beyond the reach of the average person.
Thank God for nuclear power. Clean, safe, effective. No more black lung for miners, explosions on oil rigs, power plants belching out acid rain. Remember all those?
Yes, nuclear accidents happen. Yes, they are dangerous. Yes, plants need to be safer. Everything needs to be safer. I just hope you all put on that SPF 50 when you go out in the sun. That solar radiation will get you faster than something from a power plant.
Thank you so much for this article! I also would like to echo several here who would like to know if they have been exposed to radiation and contamination from this nuclear catastrophe. I looked in your “resources” area of your website and there are no resources for people who wish to empower themselves by testing their own environment with monitors, etc… How does one best test their own garden produce for radiation or contaminates? Do you use that inspector radiation monitor that you use in the video? Or would you have to send in a sample to a laboratory? What laboratory would you suggest? Please empower us and thank you again for your work!
Two new words to help describe what is happening at SORE (San Onofre Reactor Emergency) and many other places around the World:
Nuclear Fix*
* http://is.gd/DzSrY1
The nuclear industries (aka nuclear fascists) policy of donating massive amounts of money to insure that all levels of Government support Nuclear Energy to protect their market share despite it’s enormous environmental RISK of yet another Fukushima, instead of supporting less expensive, NON RISKY Eco Friendly Solar energy.
and
Nuclear Conflict of Interest**
** http://is.gd/WiYZpz
A Nuclear Conflict of Interest happens when elected Leaders give their support to the Nuclear Industry because they have received some form of Nuclear Payback without disclosing it to the public.
Where are the references , and citations for these claims?
Michael is so right about “No Place to Hide” – Cesium detected in Canadian Quebec Maple Syrup and in New Zealand yogurt….
http://translate.google.ca/translate?hl=en&sl=ja&u=http://securitytokyo.com/&prev=/search%3Fq%3Dtokyo%2Bsecurity%26hl%3Den%26client%3Dsafari%26rls%3Den%26prmd%3Dimvnsu&sa=X&ei=NystUMmoC8iuiALqkoHQCw&ved=0CF4Q7gEwAA
http://translate.google.ca/translate?hl=en&sl=ja&u=http://securitytokyo.com/&prev=/search%3Fq%3Dtokyo%2Bsecurity%26hl%3Den%26client%3Dsafari%26rls%3Den%26prmd%3Dimvnsu&sa=X&ei=NystUMmoC8iuiALqkoHQCw&ved=0CF4Q7gEwAA
@ Ralph –
You are so right about the “unprecedented assault”. Never in the history of Mankind has the planet EVER been subjected to the ongoing radiation releases that are coming from Fukushima. We will never know how much radiation has come our way. Initially it may have been 950 terabecquerels/HOUR and now it may be “only” 10 million bq/HOUR plus the 1000 TONS or radioactive water being dumped into the ocean DAILY…!!!!!! but it is truly an UNPRECEDENTED assault.
We look forward to the Natural Solutions White Paper and we thank Michael and Denise again for the investigative reporting and bringing us all together so we can get to the TRUTH.
Re: No Place to Hide:
Thank you Michael for this excellent and comprehensive article that so clearly outlines the effect that Fukushima is having on all of us in North America.
This is a “must read”. Please – everyone – send this on to as many people as you can. Our governments are not testing our air, food or water and the mainstream media is not reporting. This deafening silence gets louder every day as the situation worsens and the contamination increases.
We are incredibly fortunate to have Michael and Denise who work tirelessly to provide a website and comment section where we can go to find out the truth and discuss it together.
Thank you for your important reporting. Here in NW NJ my radiation detector readings are still relatively low, in the .090 mSv per hour range mostly, but the heppa filter on the whole-house AC return, a week after cleaning, showed .250+
We’ve given much consideration to one issue you raise: that this assault of radiation is unprecedented, even when considering the era of above-ground testing.
The three trustees of the Natural Solutions Foundation are preparing a White Paper on Genome Disruption Syndrome, written to define the disease being caused by Fukushima (and other genome disruptors) from the viewpoint of our own expertise. Foundation President Bert Stubblebine sees it from an engineering standpoint, co-trustee Rima E Laibow MD defines the mode of operation and necessary therapy while I look at the remedies from a legal standpoint.
Our conclusion: the radiation increase you document is triggering a newly defined symptom complex that requires natural solutions to remedy the harm we are all suffering.
The White Paper should be published within the next month.
I had intended to seek relative safety in the Southern Hemisphere, if needed. Just knowing that was an option brought some level of comfort. Now, as you say, there’s no place to hide. We will have to do the best we can wherever we are located. Shelter in place, filter your water and check your food for extra radiation. It’s getting very worrisome for future generations.
Collins qualifies as the official reporter for the Apocalypse, which seems about to happen as Republicans take away protection from any industry.
My sister called me last night and asked me what are normal levels of background radiation because apparently there are helicopters flying over San Francisco with geiger counters measuring background radiation. I “think” they may be a little late on trying to get an accurate background level. ha ha. Funny they didn’t do this a year and a half ago and I find it extremely odd that Berkley wouldn’t have those numbers.
My sister said it was on the news because people were wondering why the helicopters were in the air and what they were doing. The news said “we are trying to sample normal back round radiation just in case there is a terrorist attack” WHAT??? they went on to say it has nothing to do with Fukushima. We all know better than that! I told my sister they will probably just raise the levels of what is back ground and say those are normal. Like “ya 70 is totally normal for San Francisco” Whatever! The lies keep piling up. Eventually it will all come out.
Wendy,
Most of us with kids have been following Michaels (Mr. Man hands. For those who have been with us since the beginning.) website since this all has happened. We KNOW how to protect our families. Doesn’t matter where you are, you better know what to do. We love our kids more then life itself and we are no different then you and I for one didn’t appreciate your comment. I am sure you were just thinking out loud and didn’t mean anything by it. We are a support team (family) on this site and like to help each other and stay positive as much as we possibly can.
Michael and Denise as always THANK YOU!!! and to all the other wonderful people on this site that put my mind at ease a little each day.
@Joan: Next the Hemlock Society sponsors this site so it doesn’t need to take ads anymore. 😉
The 99% VS the 1% – A battle to be fought and won with truth and unity! The people have finally started to come together. You cannot convince me that tiny Iceland can handle the “Cabal” and WE cannot!
Now is the time to join forces with those you would not normally consider. THEY win as long as they can keep us divided and focused on diversions like Left vs Right. Others have carried the load of exposing this corrupt activity for years. Now we the people NEED to come together and make it a major issue in the news and in this political cycle.
JOIN US, this is YOUR cause too (Click the “LIKE” button and spread the word): http://www.facebook.com/BankAndPoliticianFinancialCorruption
“the great tragedy of the Fukushima accident is that Japan shut down all its nuclear reactors. Even though officials have now turned two back on, the hardships and economic disruptions induced by this policy will be enormous and will dwarf any danger from the reactors themselves.”
Who is this Muller freak? “Hardship and economic disruption” dwarfs thousands (millions?) of future long, slow, and painful deaths? OK, whatever you say.
[Commenter] Michael embodies the exact problem that ails America! Whining about ADS while we are discussing the world’s biggest environmental disaster EVER? Good riddance guy, we won’t miss you. I bet you’ll be one of the first cry babies out there begging for help when the whole mess collapses.
Some of it is in the air, but some is swimming, walking and crawling!
Fukushima fish X 380 safe radioactive limits…that means the mollusks are X 3800 (ten-fold variation in the bioaccumulation hierarchy)
This was an NHK and Radio NZ story.
So now what?
Michael: I would love to be one to do testing, but 600- is a little out of our budget atm. If you ever want to donate one, I’m happy to do all the work. Everyone is harvesting their gardens now, making jams & jellies from the abundance of wild berries that grow, mushrooms are being picked, and it’s tuna season so, everyone’s busy canning those too. If I mention radiation to family or friends, they think I’m nuts. The debris are starting to wash up on the beaches as well, so there is a lot to be tested.
Wendy: It’s called a recession. Plus, after reading the article, obviously no where is safe atm. Sounds like most of the country is radiated.
The more folks with detectors the sooner the Gov’t. will have to be honest about radiation…
Micahel, with much respect, please, just enable copying. As countless industries and organizations and groups have discovered already, all forms of copy protection are inherently worthless. It takes only one person with the know-how to circumvent the protection on something; and once that person posts his single unprotected copy, the SAME amount of uncontrolled re-copying, which you fear, then proceeds to take place as if the protection never existed in the first place. Copy protection literally does nothing to stop what you’re fighting; instead it just delays the commencement of the re-copying process until the first person who can circumvent it comes along — which may mean a day, or even an hour. And that is an incredibly moot benefit compared to the much broader negative consequences copy protection has upon those without the knowledge to circumvent it, but with dire and legitimate needs to copy things — like this chap here with the medical disability. I guess this is why I have grown to hate all forms of copy protection; it is and has always been a fool’s good, the benefits of which to publishers are completely phantasmic, and yet for which the downsides to the innocent are actually quantifiable and immense.
As one example, and to prove a point, I am a webmaster, and from that alone, I immediately knew exactly what to do to my web browser to disable your anti-copying mechanism; and it worked just fine. In fact, the particular browser feature that supports it was actually disabled in mine when I came to your site (I surf with it off by default). So it wasn’t until I actually saw Jim’s comment that I realized that I had to *enable* that feature in the first place in order to even experience the inability to copy your text. Prior to doing so, I could. Which I suppose raises two points: not only has someone with the ability to defeat this stuff arrived on your site the very day of your publishing the article, but I was also someone who, because of a coincidental browser configuration, could have copied it anyway, without my knowledge, and even without knowing that any copy protection mechanism was actually there in the first place.
Anyway, FWIW, I will not repost your article elsewhere, and I also will not divulge the means of enabling copying. I don’t want to appear hostile to you, and discredit my plea in your eyes. I would much rather offer this debate instead, hoping you might see that while this stuff is well-intentioned, it is ultimately folly that cannot possibly protect you, while also harming those who mean you no harm.
My humble advice (a.k.a. an idea): dump the anti-copying stuff, and use a server-side include (or some equivalent in your CMS) to make a modest banner appear in each of your articles. Make it say something like: “This web site depends upon ad-supported visits to survive. If you would like to share this article, PLEASE do not simply copy and paste its text into other forums. Instead, post only its title, its link, and a brief excerpt of your choice. This truly makes a tremendous difference to us, and will ensure we can continue operating.” I would think that with this — i.e. asking nicely, 90% of people would oblige. All you’d need to do is make sure the notice is attention-getting. It could appear at the top and bottom of each article’s table body, for instance. Maybe there could even be a third iteration as an inset in the middle of each article. Whatever. But most people are honest and would oblige, I’d think; and you’d no longer be punishing the innocent this way.
Sorry for the long rant, but I suppose its length is born out of my frustration with this stuff. It never, ever does anything but hurt the wrong people, and I suppose it’s doubly frustrating to me to see it being resorted to amongst the alternative media, as opposed to by the usual brainless corporate suits in the mainstream (who’s crap is so useless it’s not worth copying anyway — oh, the irony). Your site, and especially articles like this, are wonderful humanitarian services, and the research and effort you’ve put into this article alone is jaw-dropping, so I can’t thank you enough for it. But as much as I can sympathize with your feelings after seeing your work stolen (as someone with his own experiences in that right), I can’t bite my tongue here and NOT argue the reality of it.
Thanks for listening.
Our political leadership is more interested in staying in power then the consequences of doing nothing following Fukushima.
May Botox Boxer, Prune face Pelosi, and Childish Obama be held accountable for DOING NOTHING.
I’m in Wisconsin. Where are the graphs for my state?
@SBLocal: Once we have completed the distribution of this article, we will create a list of potential hair sample analysis labs. I agree that sending samples to the same lab would be a plus IF that lab were 100% accurate. But since we don’t know that – that is unless a lab contacts us and presents a great case as to why their quality control and quality assurance is the best – use of multiple labs shouldn’t change the fact that people will have their hair carefully analyzed.
@Michael: Since when are ads, ads that help pay for the work, bad? Do you turn off the television, avoid radio, not go to the ball park and never drive your vehicle down the street because of ads? I think not. However, it may be helpful if you would donate to the site more than just hot air.
@Jim: We don’t allow copy and pasting for the simple reason people take our material and repost it in its entirety with no credit and no links and then make money of our work through ads on their websites, some of which we want no association with. That is theft and hurts our ability to do this work because, just like most sites on the Internet, we need people to come to our site to read our material in order for us to make the small amounts of money we do from advertisements and donations. To share this material, simply write the name of the article, copy its link and send it to whomever you want. We apologize if it is hard for folks with disabilities to access this information but we have no more effective way of sharing it than to post it as we have.
@Colleen: We really want someone to step up in Oregon so we can establish a Radiation Station there in order to keep you informed of not just the air readings, but of the rain detections and sampling and testing of Fukushima debris washing up on the coast. We also want food reading from grub grown there as the Cascades in Oregon were hit worse than anywhere else in North America by the initial blast of fallout. Maybe that person is you Colleen if you can spring for the $600 or so it will cost to get an Inspector Alert nuclear radiation monitor, webcam and computer to begin live streaming.
Are mothers with young children still living on the West Coast in California, Oregon, and Washington? What are you, nuts?
Where can I send my hair sample? (if everyone uses the same lab the results are more credible). This is magnificent work! Thanks to you both.
Michael,The blaring no opt-out corporate ads that play right after your page has loaded and intermittently thereafter, are a Major Distraction to your obvious credibility and the value of your message. I frequently listen to you on Rense and have visited your site a number of times, but I will not be visiting the site any more!
LOTS more comments about SORE (San Onofre Reactor Emergency) here:
Also there is a new “whistle blower” posting comments about SORE (San Onofre Reactor Emergency) here http://is.gd/BqPIbB
and
If San Onofre nuclear plant is restarted, who pays?
http://is.gd/VpaCpR
and
Nuclear Regulatory Commission may study power plant health risks
http://is.gd/SY6iPU
Will this also happen in the USA if we have a Fukushima?
Elderly Evacuees from Futaba-machi Living in School Bldg in Saitama Will Be Made to Pay for Their Boxed Meals, Starting September 1st
http://is.gd/QmifHR
snip
Just remember that this country (Japan) is still the third largest economy in the world. But after more than 17 months since the earthquake/tsunami/nuclear accident, hardly anyone cares that these people are still living in school classrooms. It’s worse than not caring, because now they will charge for the bento (meal in a box), which will cost these elderly residents 30,000 to 40,000 yen per person per month (US$381 to 508 per person per month).
Where do they have such money? No one cares. The evacuees from the same town, Futaba-machi, who have moved to temporary housing and other rental properties, have complained that they are not getting free meals, so everyone gotta pay, to be fair.
Why do you make the page such that it cannot be copied or pasted? those of us with reading difficulty need text to speech programs, often due to MS,and other neurological illnesses. So why do you make it impossible for us to get the information? Donations to your oganization are a waste for those of us that cannot access the information, for some strange reason you want to protect your information??? perhaps! give us a break let us that need assistance in reading use natural reader or the other programs that allow us to use text to speech.
Perhaps this is all for the best….The human race continually causes ruination and destruction or allows predatory psychopaths to seize power and bring about such damage every generation. Such permanent design flaws in a species can never be fixed or corrected, so as long as there are humans, there will be tragedies such as this.
Great article!
My 2 year old daughter is, unfortunately, the perfect candidate. She has not has her hair cut yet whereas her 4 year old brother has had it cut many times. We are in for the tests. The next step is where to find the hair analysis testing agencies. We live in Korea at the moment and are actively looking.
Suggestions?
Recently, I have been in touch with my MLA in Saskatchewan who then forwarded it to the Emergency Management Organization who told me it is the responsibility of the Federal Government to conduct testing for fallout. Thank you for your concerns. You can find their link…….lies, lies and more lies…..
This is so frustrating. Since last year, I have not found a single article about radiation in Oregon. Oregon had 37 1/2″ of rain in March of this year. That is in 1 month, how much rain has California had in 1 year? Not nearly that amount I guarantee. So why are all these tests being done in California and not Oregon? I live on the coast with 2 small children and, I have no clue as to if we are safe here. Would someone please do tests in the Oregon coast? Please?